If you’re losing your mental edge, the real culprit might not be aging; it might be a hormone no one bothered to test. Many men experiencing brain fog, slowed thinking, or mild forgetfulness are often told it’s “just aging.” Yet emerging human research 1 shows that low testosterone, not age alone, may be a driving factor in cognitive decline. Because most neurologists do not routinely evaluate hormones, many men receive a “you’re fine” diagnosis despite apparent symptoms.
Before you accept brain fog and memory lapses as aging, consider the role testosterone plays in brain health. At Vita Bella, we help men identify hormonal issues that traditional neurology overlooks. Your cognition depends on more than scans and tests; it depends on balanced hormones. Learn how optimizing testosterone can support clarity, focus, and long-term protection.
Why Doctors Overlook the Hormonal Cause, Especially Low Testosterone?
Neurologists rely heavily on strict diagnostic cutoffs for mild cognitive impairment (MCI) or dementia. If a man scores above the impairment threshold, he is often reassured that his symptoms are normal aging. However, testosterone declines naturally after age 40, and many men fall into the low range without meeting criteria for hypogonadism. Evidence 1 shows that lower testosterone levels in middle-aged and older men significantly correlate with a higher incidence of cognitive decline and dementia.
Moreover, studies 2 on men reveal dramatically higher rates of mood changes, dementia, cognitive impairment, and Alzheimer's disease, highlighting how sharply the brain responds to reduced testosterone. When testosterone drops rapidly, cognition often declines in parallel. This reinforces the idea that hormone imbalance is biologically significant even before dementia emerges.
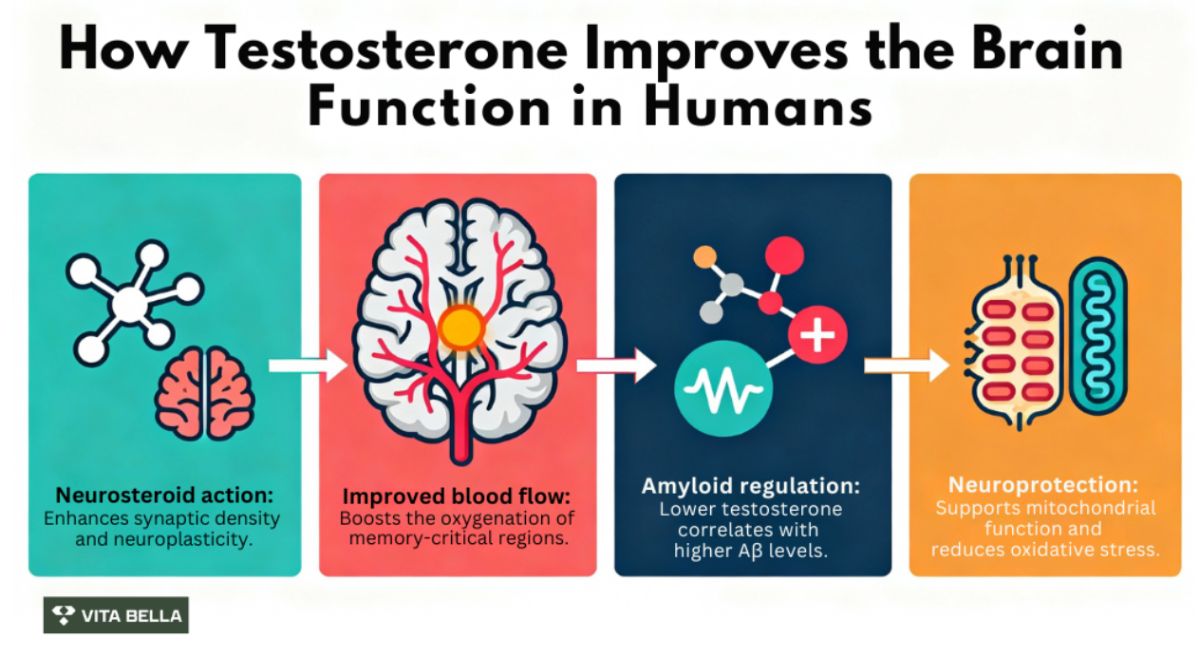
Can Testosterone Function as a Neurosteroid to Support the Aging Brain?
Testosterone acts as a neurosteroid, meaning it influences neurons directly. It supports synaptic activity, neuroplasticity, and neurotransmitter balance. In a 14-year longitudinal study 3, older men with higher free testosterone showed better regional cerebral blood flow in memory-critical areas such as the hippocampus and anterior cingulate cortex. Improved blood flow supports oxygen delivery, nutrient transport, and removal of metabolic waste, key functions that protect cognition.
A study 4 explains that androgens can enhance cognitive function via other substances and related pathways. As various carriers enhance the brain's sensitivity to testosterone, carriers are more resilient to the effects of aging brought on by decreasing testosterone levels, which cause cognitive impairment. Furthermore, it has been demonstrated that the brain's aging endothelial cells might accelerate the aging of neurons.
Testosterone stimulates the production of aged vascular endothelium cells by increasing eNOS activity. This prevents endothelial cell aging, which in turn prevents neuronal aging and slows cognitive decline. Testosterone supplementation has clear neuroprotective benefits, and testosterone levels are linked to cognitive decline. It has been demonstrated that taking 50–100 mg of testosterone daily improves verbal memory, overall cognition, and depressive symptoms.
What Is the Testosterone–Amyloid-β Connection?
Amyloid-β (Aβ) accumulation is strongly associated with Alzheimer’s disease. Human observational research 5 demonstrates that men with lower testosterone often have higher levels of circulating amyloid-β. In men presenting with memory complaints, those with low free testosterone showed significantly elevated plasma Aβ1-42 concentrations compared to men with healthier hormone levels. This suggests testosterone may help regulate Aβ production or clearance.
Further clinical endocrine research 6 shows that androgen deficiency correlates with increased Aβ plaque formation and impaired synaptic function in humans. Although more research is needed, early findings strongly imply that testosterone acts as a protective factor against the pathways involved in Alzheimer’s disease.
What the Human Evidence Shows about Testosterone-Dementia?
Studies link low free testosterone to reduced brain blood flow, poorer memory performance, and elevated amyloid-β levels, key markers associated with Alzheimer’s progression. These findings highlight testosterone as a crucial, modifiable factor in men’s long-term brain health. Studies consistently demonstrate the following patterns:
Men with low testosterone have higher rates of cognitive decline and dementia.
Higher free testosterone predicts better brain perfusion and slower cognitive aging.
Low testosterone correlates with higher amyloid-β levels in men with memory issues.
Testosterone therapy in small human trials improved visuospatial function and quality of life in men with early Alzheimer’s symptoms.
Why Neurologists Miss Testosterone in Cognitive Complaints?
Because cognitive symptoms often appear subtle and men may mask them well, neurologists depend on cognitive testing to identify diagnosable impairment. But many men with low testosterone remain above the impairment threshold while still experiencing memory lapses, fogginess, slowed recall, and reduced concentration. These symptoms may not qualify as MCI, but they are not “normal.” Low testosterone can produce cognitive changes that mimic early dementia, yet are reversible with appropriate hormonal evaluation.
Most neurologists do not routinely test testosterone unless sexual symptoms are reported, making cognitive-related hormonal decline easy to miss. This creates a diagnostic gap: men with hormone-driven cognitive impairment are told they are aging normally, delaying interventions that could improve brain function and prevent decline.
Practical Steps for Men and Clinicians
Testing provides a more complete picture of hormone-brain function. Evaluating testosterone early can help men avoid unnecessary worry, misdiagnosis, or delayed treatment for preventable hormone-related decline. Men reporting cognitive changes should request:
Total testosterone
Free testosterone
SHBG (sex hormone–binding globulin)
Estradiol (since some testosterone converts to estrogen in the brain)
Move Beyond Normal Aging, Optimize Your Health with Vita Bella
Are you frustrated by brain fog but told your tests are “normal”? Traditional evaluations often ignore testosterone, a key player in brain health. Low testosterone elevates dementia risk and accelerates cognitive decline. Missing this hormone means missing a significant, reversible cause of symptoms. Your mind deserves a real explanation, not a dismissal disguised as reassurance.
Vita Bella takes a deeper approach to male cognitive health. We integrate hormonal testing with scientific strategies for brain optimization. Testosterone improves memory, focus, and mental stamina from the inside out. Stop settling, start strengthening your brain with Vita Bella. Take the first step toward clarity with a solution designed specifically for men.

FAQs
Can low testosterone contribute to cognitive decline in men?
Yes, multiple human studies show that men with low testosterone levels face a higher risk of cognitive decline, impaired memory, and dementia. Low testosterone can reduce brain blood flow, weaken neuronal function, and elevate amyloid-β levels, key contributors to Alzheimer’s disease. This makes testosterone a critical, modifiable factor in long-term male brain health.
Do neurologists often miss hormonal causes of brain fog in men?
Yes, neurologists typically diagnose based on cognitive test thresholds, so many men with low testosterone fall outside “impairment” ranges and are labeled normal. Because hormone testing isn’t routinely performed, testosterone-driven brain fog and slowed thinking often go unnoticed, leading to misdiagnosis and delayed treatment for a reversible, hormone-related issue.
Can testosterone improve brain function when levels are clinically low?
Yes, Testosterone acts as a neurosteroid, supporting blood flow, synaptic activity, and cognitive processing. When men with clinically low levels receive appropriate treatment, studies show improvements in attention, memory, and overall mental clarity. While it’s not a cure-all, restoring testosterone can enhance brain performance and may reduce long-term cognitive risk.
Is there a link between testosterone levels and Alzheimer-related changes?
Yes, human research demonstrates that men with lower testosterone often exhibit higher amyloid-β accumulation, a hallmark of Alzheimer’s disease. Low testosterone may weaken the brain’s ability to regulate and clear these proteins. Because this mechanism appears early, monitoring testosterone could help identify men at elevated neurological risk long before dementia fully develops.
References:
Yeap, B. B., & Flicker, L. (2022). Testosterone, cognitive decline and dementia in ageing men. Reviews in Endocrine and Metabolic Disorders, 23(6), 1243–1257. https://doi.org/10.1007/s11154-022-09728-7
Lu, P. H., Masterman, D. A., & Mulnard, R. (2006). Effects of testosterone on cognition and mood in male patients with mild Alzheimer disease and healthy elderly men. Archives of Neurology, 63(2), 177-185. https://doi.org/10.1001/archneur.63.2.177
Moffat, S. D., & Resnick, S. M. (2007). Long-term measures of free testosterone predict regional cerebral blood flow patterns in elderly men. Neurobiology of Aging, 28(6), 914-920. https://doi.org/10.1016/j.neurobiolaging.2006.04.001
Cai, Z., & Li, H. (2020). An updated review: Androgens and cognitive impairment in older men. Frontiers in Endocrinology, 11, Article 586909. https://doi.org/10.3389/fendo.2020.586909
Verdile, G., Laws, S. M., Henley, D., Ames, D. J., Bush, A. I., Ellis, K. A., Faux, N. G., Gupta, V. B., Li, Q.-X., Masters, C. L., Pike, K. E., Rowe, C. C., Szoeke, C. E. I., Taddei, K., Villemagne, V. L., & Martins, R. N. (2014). Associations between gonadotropins, testosterone and β-amyloid in men at risk of Alzheimer’s disease. Molecular Psychiatry, 19(1), 69-75. https://doi.org/10.1038/mp.2012.147
Lei, Y., & Zhou, R. (2018). Effects of androgens on the amyloid-β protein in Alzheimer’s disease. Endocrinology, 159(12), 3885-3894. https://doi.org/10.1210/en.2018-00660





















